
Diffusion is the primary mechanism in dispersion, although it is driven by convection in some cases. In convection, variation in velocity between paths of flow in the bulk medium facilitates the equal distribution of dispersed material in the medium. This results in an equal dispersion of particles in the medium. The difference in engagement between dispersed material and the bulk medium creates a concentration gradient that drives the medium's dispersion. Through molecular diffusion phenomena, dispersion occurs through different concentrations of the media introduced throughout the bulk medium. This whole process is aided by molecular diffusion and convection. A new line of interaction between the dispersion medium's inner surface and the outer surface of dispersed particles is created. The agglomerated particles get separated from each other. Because of this nature, these are known as Associated Colloids and also famous as a micelle. However, when a suitable condition like in higher concentration, they show the behavior of colloidal particles. This arrangement is popular as Macromolecular Colloids.Įxample: Enzymes, cellulose, and proteins are naturally formed macromolecular colloids, but rubber and polythene are synthesized.īasically, these are behaving like strong electrolytes. However, in a suitable solvent, these form solutions whose size may or may not remain in the colloidal range. 
After the separation of the particles, the colloid constitutes several atoms and molecules known as multimolecular colloids.Įxample: Hundreds of Sulfur molecules are held together by van der Waals force and form Sulfur Solution. Usually, their size varies from 1-1000nm. When a substance gets dissolved in a dispersion medium, it gets separated into several smaller molecules of different sizes. Aerosols are small particles of liquid or solid dispersed in a gas, e.g., smoke, fog, mist, etc.ĭepending upon the minute particles' nature of the dispersed phase, the colloids are mainly differentiated into three types: Foam is formed when gas particles get trapped in a liquid or solid, e.g., soap in water. An emulsion is a colloidal suspension of two drinks, e.g., milk. A colloidal suspension of solid particles in a liquid is called a sol, e.g., Ruby glass. These include sol, emulsion, foam, and aerosol. Fast sedimentation of the dispersed phase is observed owing to the coarse dispersions, mainly due to gravity.ĭispersed Phase: Classification and Typesīased on the type of dispersed phase, colloids can be classified into various types. These are heterogeneous dispersed systems. The second type of dispersion medium is of the coarse kind. Examples of molecular dispersion are air (consisting of various gasses like nitrogen and oxygen), electrolytes, and alloys. The dispersed phase is homogeneously distributed in the dispersion medium. Molecular dispersions are dedicated solutions to a solute phase in the solvent. There are two types of dispersion systems, Molecular Dispersions and Coarse Dispersions. Examples of the dispersed phase include dust in the air, whereas that of dispersion medium includes water in milk. The dispersed phase is known as the internal phase, whereas the dispersion medium is called the external phase. The dispersion medium is the continuous phase of a colloid, and it distributes throughout the dispersion medium. It is one of the two stages of a colloid. The dispersed phase of dispersion is the discontinuous phase that distributes throughout the dispersion medium. Shaving creams contain tiny airdrops of water dispersed in the air.ĭifference Between Dispersed Phase and Dispersed Medium Milk, which we most probably use daily, is made up of oil drops dispersed in water. There are various everyday life examples of dispersed media. Precisely, they can be a liquid, a solid or a gas. These two media can be different in properties altogether. A dispersed medium consists of two media that do not mix. The stage with finely differentiated particles is called the dispersed phase. One step possesses finely differentiated particles suspended in a continuous medium. 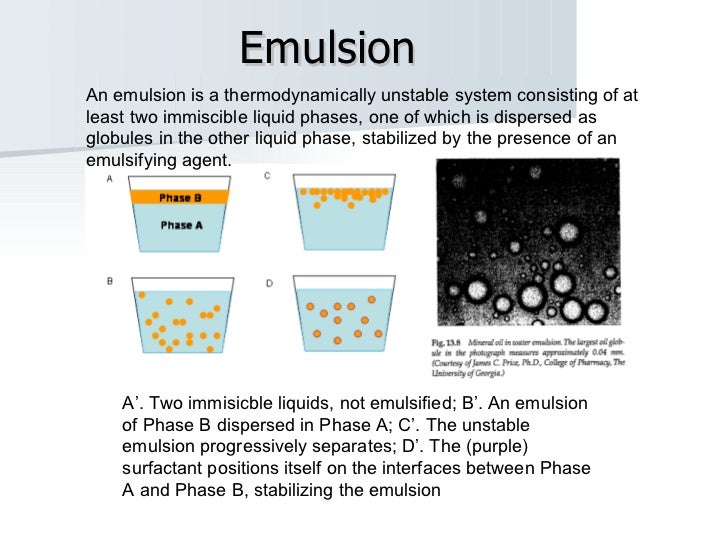
Water is considered a dispersed medium in the above criteria, and starch or ink can be regarded as a dispersed phase. Water in starch or water in ink is an example of a heterogeneous mixture. A colloid solution does not possess a uniform mixture. That is, the particles of the dispersed phase are present in the particles of the dispersed medium. In this solution, a dispersed phase is suspended in the dispersed medium. To understand the dispersed phase, we need to understand what a colloid is.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
